Science of Synthesis Advances in Organoboron Chemistry towards Organic Synthesis – Review on borylative ring-closing reactions - Thieme Chemistry - Georg Thieme Verlag KG

Sequential Ring-Opening and Ring-Closing Reactions for Converting para-Substituted Pyridines into meta-Substituted Anilines | Organic Letters
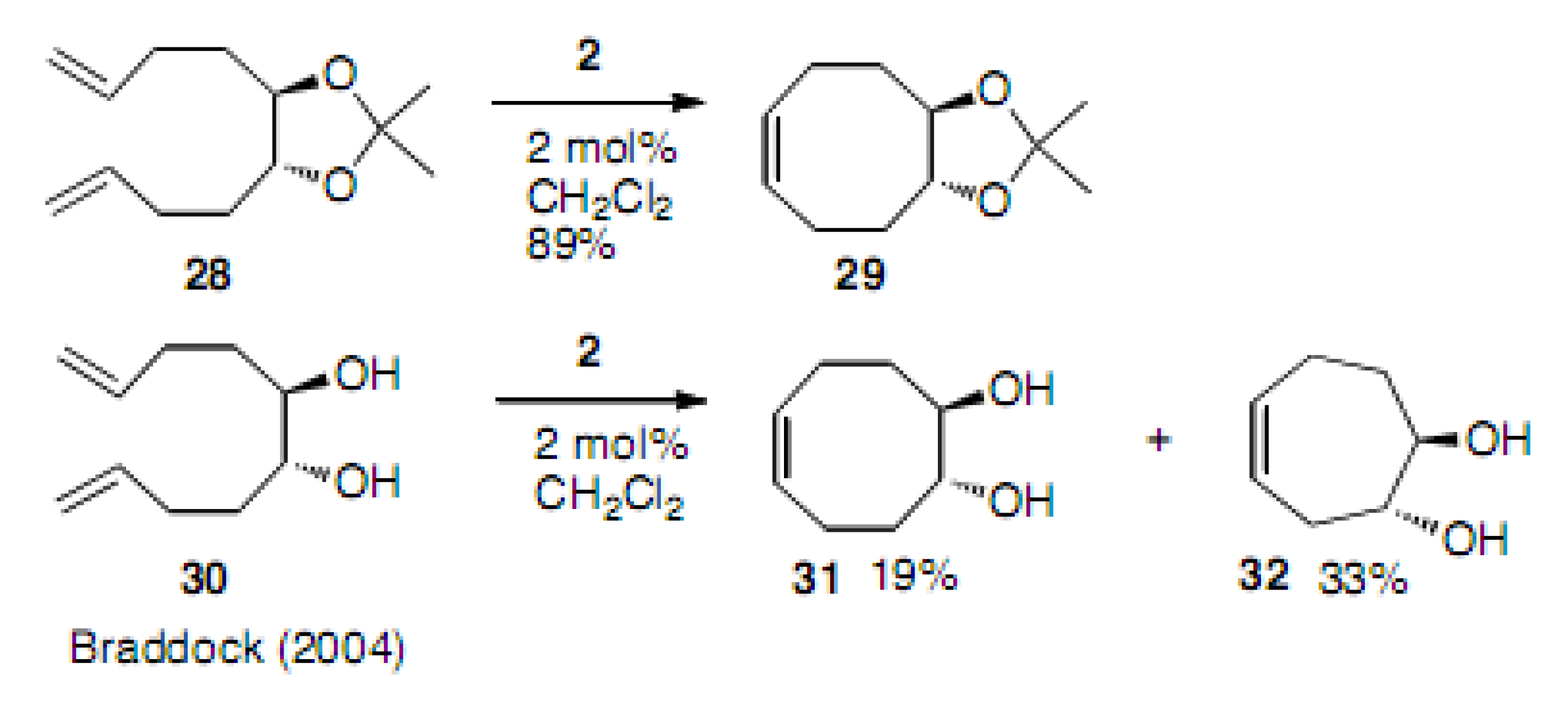
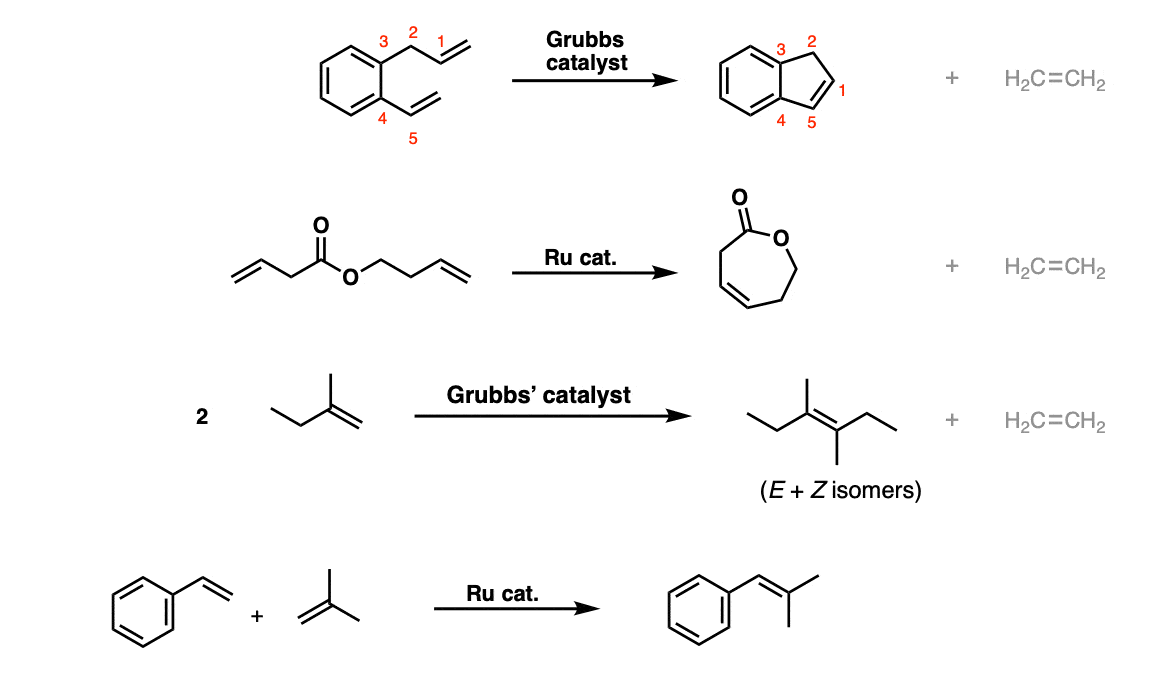
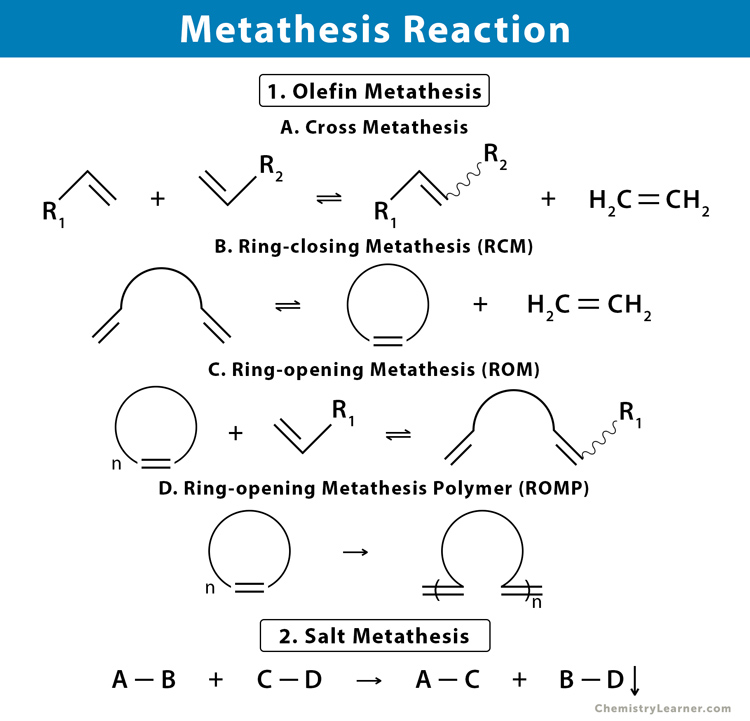
Molecules | Free Full-Text | Construction of Eight-Membered Carbocycles with Trisubstituted Double Bonds Using the Ring Closing Metathesis Reaction
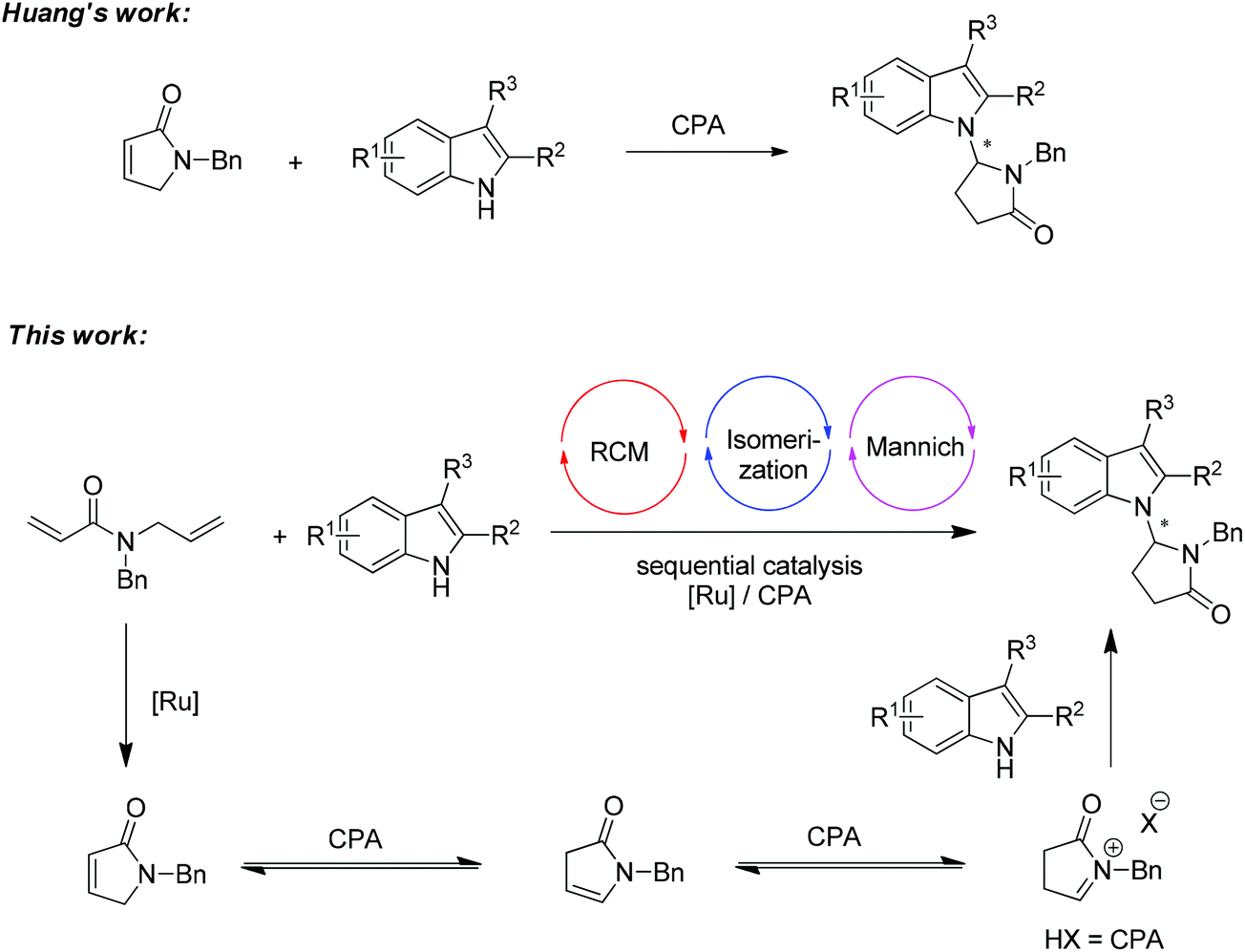
N -alkylation of indole via ring-closing metathesis/isomerization/Mannich cascade under ruthenium/chiral phosphoric acid sequential catalysis - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C3QO00008G

Sequential Ring-Opening and Ring-Closing Reactions for Converting para-Substituted Pyridines into meta-Substituted Anilines | Organic Letters
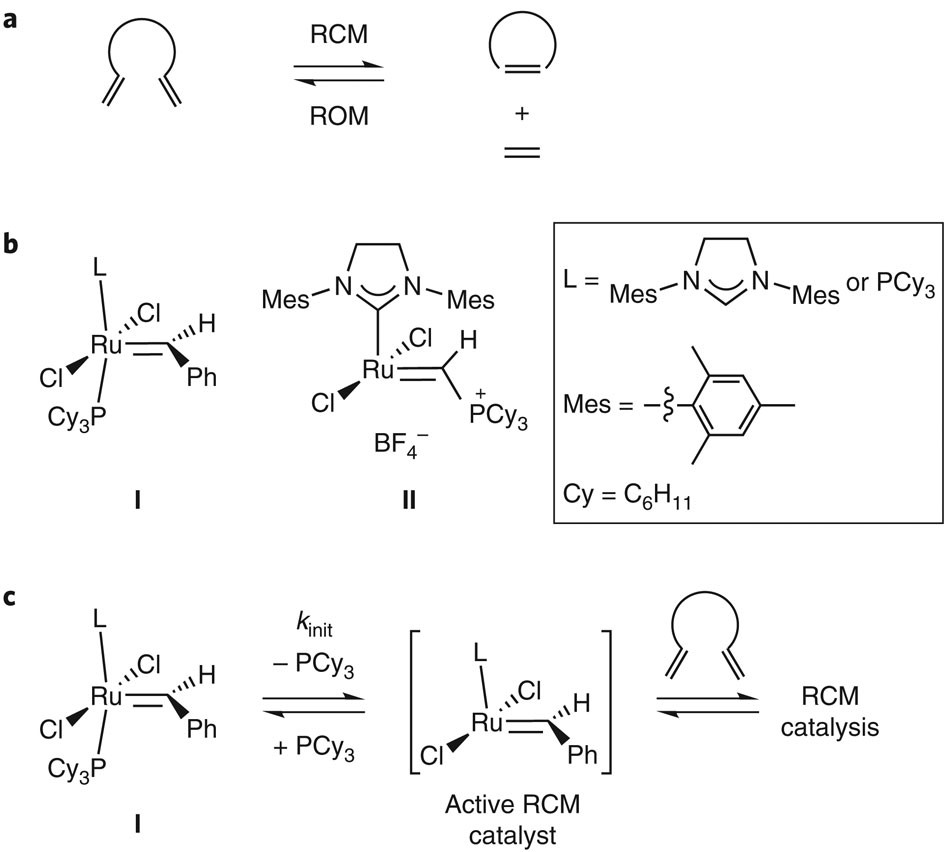
Mechanistic insights into the ruthenium-catalysed diene ring-closing metathesis reaction | Nature Chemistry

Tandem halogenation/Michael-initiated ring-closing reaction of α,β-unsaturated nitriles and activated methylene compounds: one-pot diastereoselective synthesis of functionalized cyclopropanes - Organic & Biomolecular Chemistry (RSC Publishing)

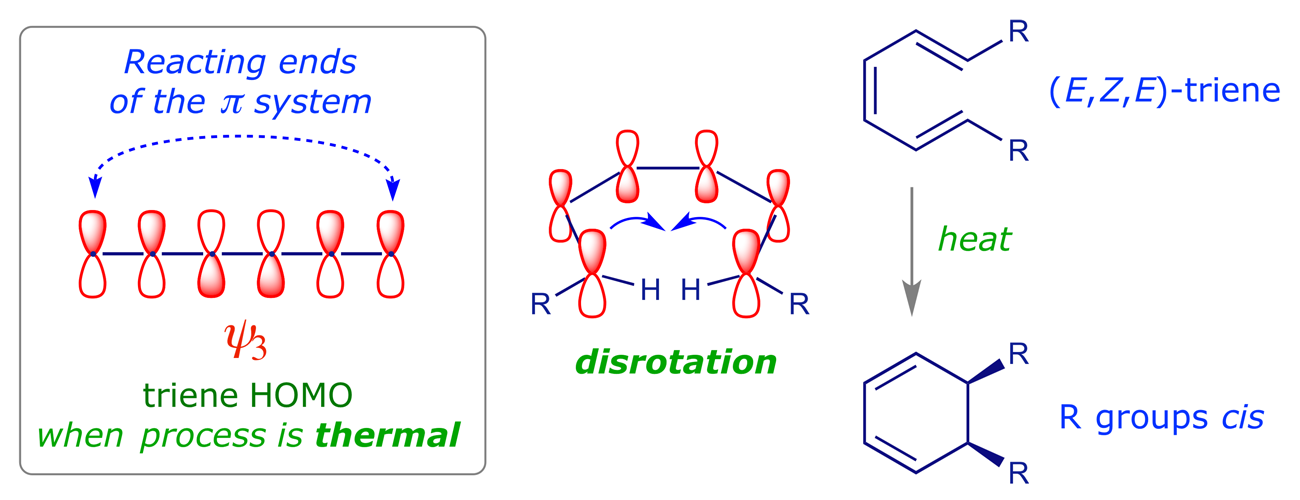
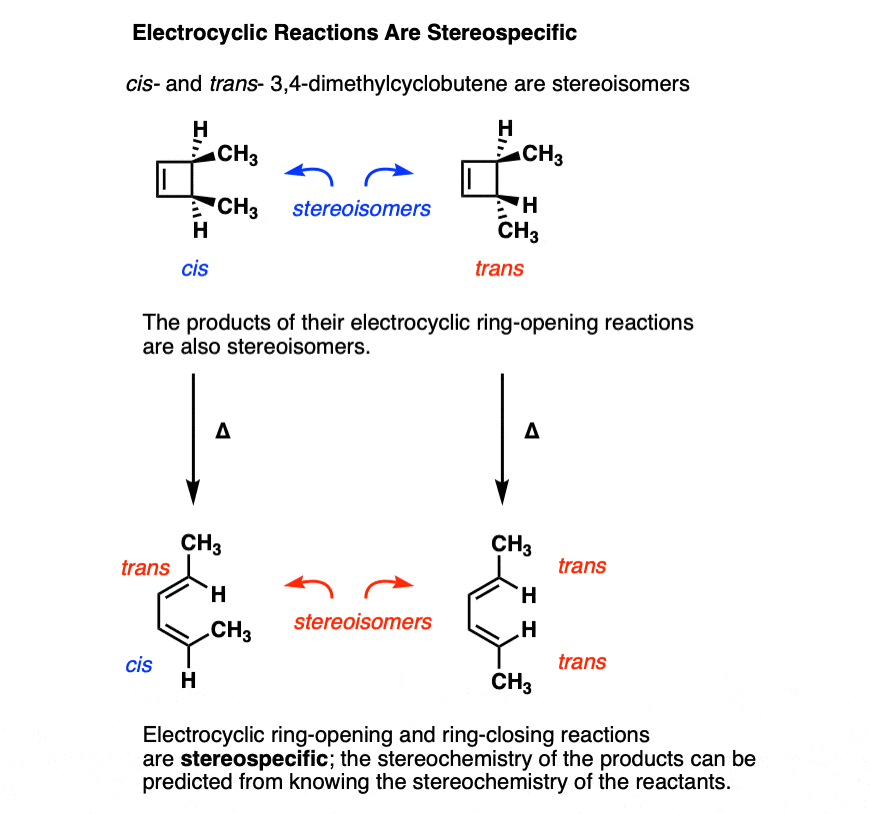
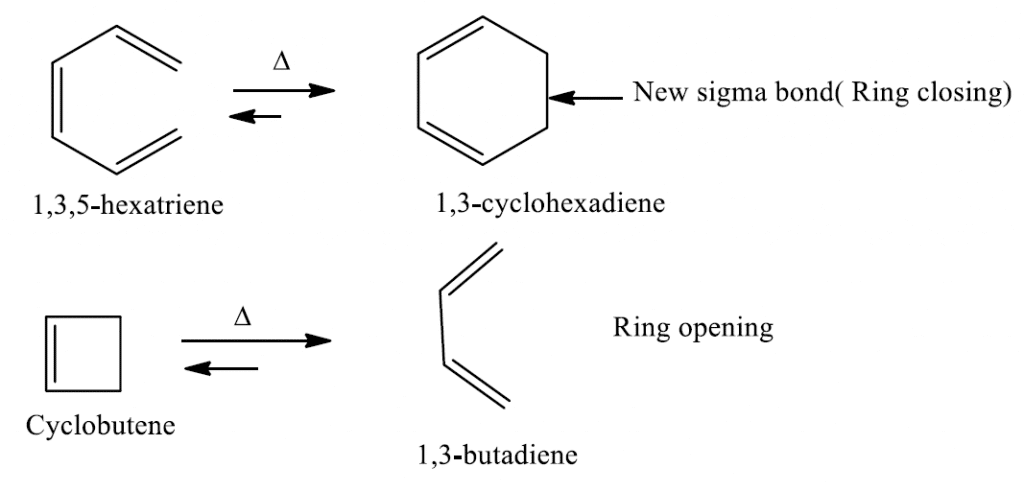
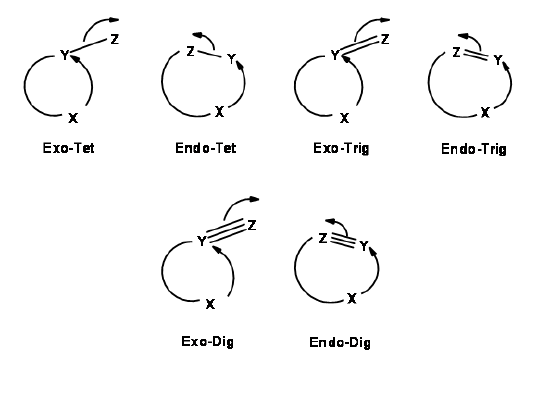
2E,4Z,6Z,8E)-2,4,6,8-Decatetraene has been cyclized to give 7,8- dimethyl-1,3,5-cyclooctatriene. Predict the manner of ring-closure-conrotatory or disrotatory-for both thermal and photochemical reactions, and predict the stereochemistry of the product ...

Michael–Michael Ring-Closure Reactions for a Dihapto-Coordinated Naphthalene Complex of Molybdenum | Organometallics