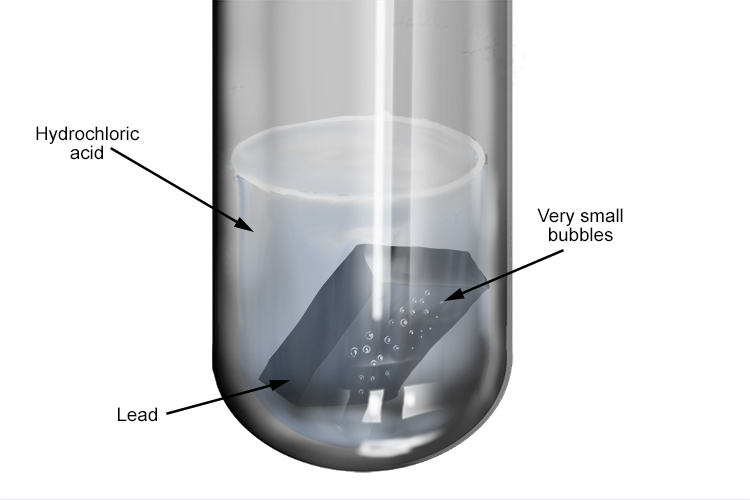
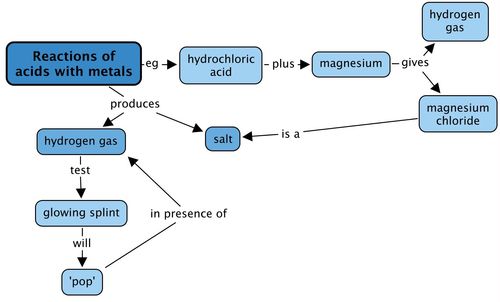
Experiment: The reaction of metals with dilute Hydrochloric acid - Free ZIMSEC & Cambridge Revision Notes

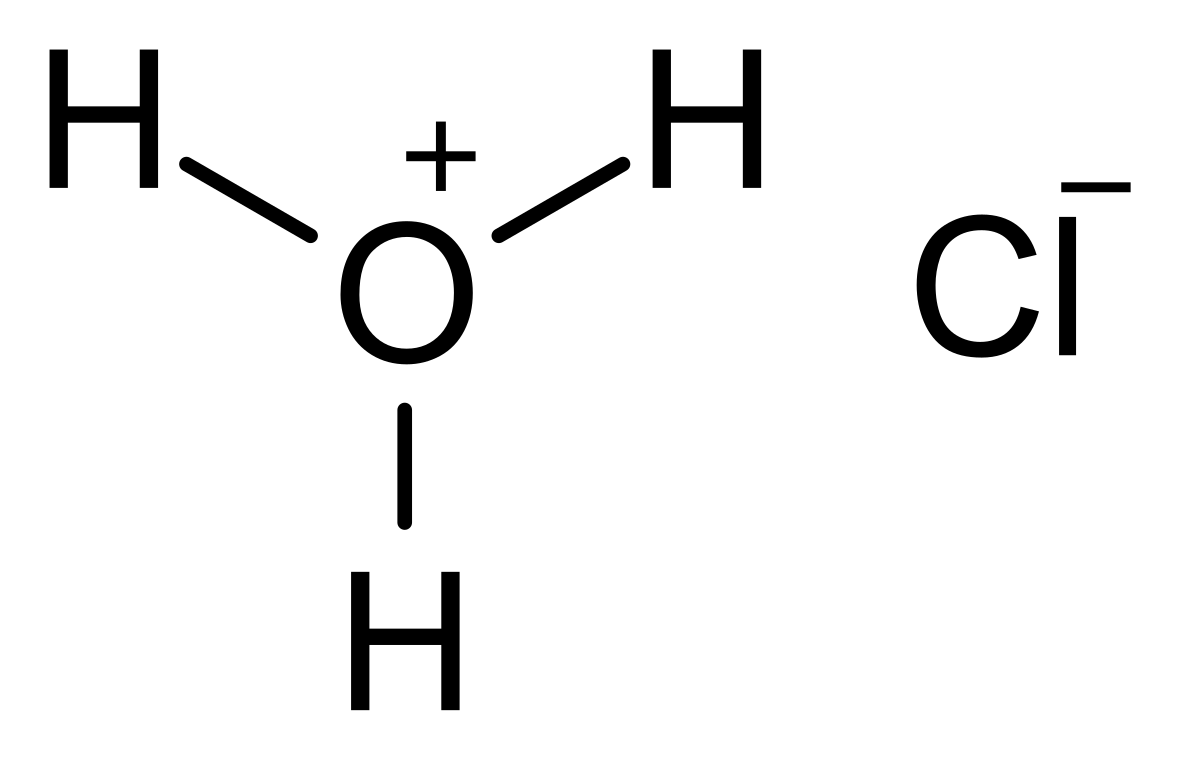
SOLVED: You set up a reaction with Gallium metal in excess hydrochloric acid at 28 degrees Celsius It reacts in the following: 2Ga(s)+6HCIaq)-2GaClylaq)+3H2lg) You collect 160 mLs and 0.966 atm of hydrogen